Imaging Voltage in Complete Neuronal Networks Within Patterned Microislands Reveals Preferential Wiring of Excitatory Hippocampal Neurons
Alison S Walker, Benjamin K Raliski, Dat Vinh Nguyen, Patrick Zhang, Kate Sanders, Kaveh Karbasi, Evan W Miller
Frontiers in Neuroscience, 2021.
[PDF]
Abstract
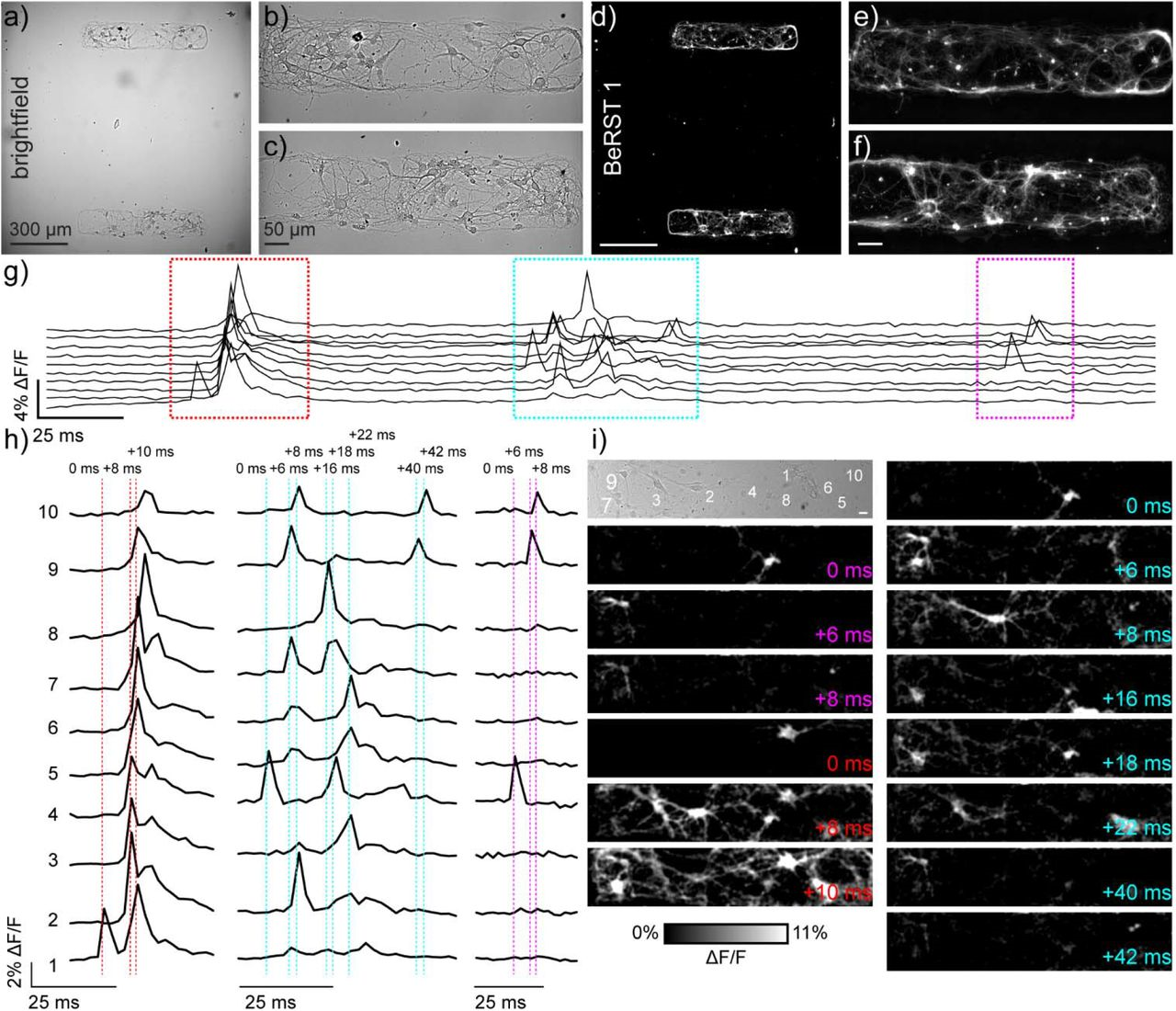
Voltage imaging with fluorescent dyes affords the opportunity to map neuronal activity in both time and space. One limitation to imaging is the inability to image complete neuronal networks: some fraction of cells remains outside of the observation window. Here, we combine voltage imaging, post hoc immunocytochemistry, and patterned microisland hippocampal culture to provide imaging of complete neuronal networks. The patterned microislands completely fill the field of view of our high-speed (500 Hz) camera, enabling reconstruction of the spiking patterns of every single neuron in the network. Cultures raised on microislands develop similarly to neurons grown on coverslips and display similar composition of inhibitory and excitatory cell types. The principal excitatory cell types (CA1, CA3, and dentate granule cells, or DGC) are also present in similar proportions in both preparations. We calculate the likelihood that action potential firing in one neuron to trigger action potential firing in a downstream neuron in a spontaneously active network to construct a functional connection map of these neuronal ensembles. Importantly, this functional map indicates preferential connectivity between DGC and CA3 neurons and between CA3 and CA1 neurons, mimicking the neuronal circuitry of the intact hippocampus. We envision that patterned microislands, in combination with voltage imaging and methods to classify cell types, will be a powerful method for exploring neuronal function in both healthy and disease states. Additionally, because the entire neuronal network is sampled simultaneously, this strategy has the power to go further, revealing all functional connections between all cell types.
